Core Technologies Column
Cryo EM at UF Reaches 3.87 Angstrom Resolution
Cryo EM at UF Reaches 3.87 Angstrom Resolution
The ICBR recently completed several upgrades to the cryoEM facility to enhance the performance of the FEI F20 FEG transmission electron microscope. Specifically, sound dampening walls and a dedicated HVAC system that could deliver sufficient cooling with minimal air current and temperature fluctuations better than +/- 1° F were installed. IFAS and Microbiology and Cell Sciences also provided funding to update existing instrument software and ancillary equipment during the process.
To evaluate the impact of these significant improvements biological specimens from the laboratory of Mavis Agbandje-McKenna, Ph.D., were used to compare image data from pre- and post-renovations and upgrades. The ICBR EM Core collected cryoEM data on several of her well characterized virus samples for comparison to earlier data gathered prior to our infrastructure improvements. Preliminary results show that the renovations and upgrades to the ICBR F20 system have greatly improved resolution. Collected image data results in the construction of high resolution 3D structures that allow the McKenna lab to model crystallographic data within the electron densities being generated to better than 4 Angstrom (Å) resolution. [An angstrom in one billionth of a meter! The point of a needle is about 1 million Angstroms in diameter!). This enables individual amino acids, on the surface of the virus structure, to be identified that are involved in antibody or cellular receptor binding. The information obtained aids the McKenna lab in the engineering of new AAV variants with improved antibody escape and tissue specific tropism properties. This in turn improves the efficacy of the AAV gene delivery vectors that her lab is developing.
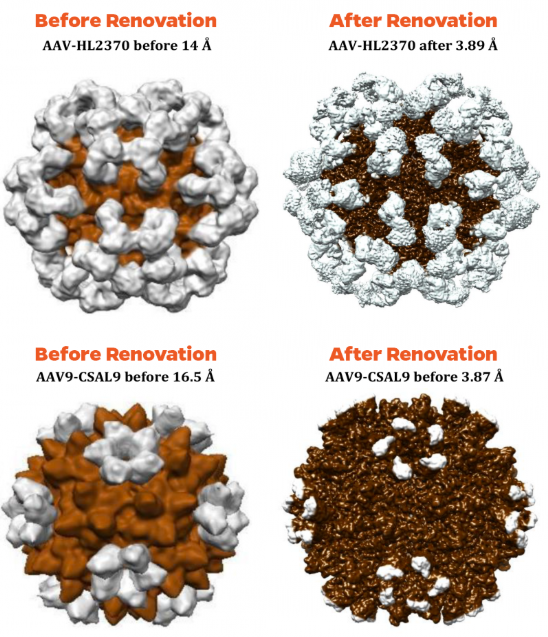
At this point, three structures have been solved using this new technology. The first was Spiroplasma Virus 4, originally published in 1996 by McKenna at 27 Å resolution, and reconstructed to 6 Å last year with our DED prior to the additional room renovations. It has now been solved to 4.1 Å demonstrating the clear improvements to the laboratory environment. Subsequent efforts have yielded models of Adeno Associated Virus (AAV) 9 with antibody HL2370, previously solved by the McKenna lab prior to the DED and renovations, to 14 Å, and AAV9 with receptor CSAL9 to 16.5 Å. Post renovations these structures are now resolved at 3.89 Å and 3.87 Å, respectively, indicating a substantial improvement in resolution due to laboratory renovations.
To Learn More:
Optimization of the data acquisition is still ongoing, as is how to best facilitate helping researchers effectively analyze the large amounts of data generated from such projects. We are currently conducting consultations on an individual basis as we gear up to offer this as a normal service. If you are interested in utilizing the cryoEM or any other EM services please contact Paul Chipman at pchipman@ufl.edu.